A. Ashraf, M. Islam, M. Khalid, A. P. Davis, M. T. Ahsan, M. Yaqub, A. Syed, A. M. Elgorban, A. H. Bahkali, and Z. Shafiq
Abstract
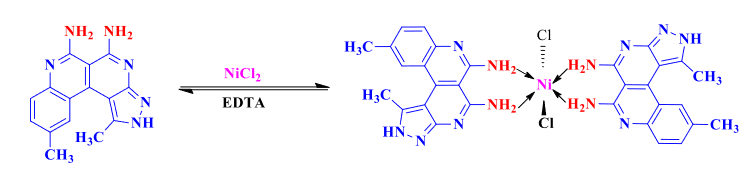
Highly selective and sensitive 2,7-naphthyridine based colorimetric and fluorescence “Turn Off” chemosensors (L1-L4) for detection of Ni2+ in aqueous media are reported. The receptors (L1-L4) showed a distinct color change from yellow to red by addition of Ni2+ with spectral changes in bands at 535–550 nm. The changes are reversible and pH independent. The detection limits for Ni2+ by (L1-L4) are in the range of 0.2–0.5 µM by UV–Visible data and 0.040–0.47 µM by fluorescence data, which is lower than the permissible value of Ni2+ (1.2 µM) in drinking water defined by EPA. The binding stoichiometries of L1-L4 for Ni2+ were found to be 2:1 through Job’s plot and ESI–MS analysis. Moreover the receptors can be used to quantify Ni2+ in real water samples. Formation of test strips by the dip-stick method increases the practical applicability of the Ni2+ test for “in-the-field” measurements. DFT calculations and AIM analyses supported the experimentally determined 2:1 stoichiometries of complexation. TD-DFT calculations were performed which showed slightly decreased FMO energy gaps due to ligand–metal charge transfer (LMCT).