As synthetic supramolecular chemists, we aim to design and construct functional molecules, while studying the intermolecular interactions which underlie their behaviour. We are especially interested in making molecules with functions that are genuinely useful and lead to real-world applications. Currently we focus mainly on two areas, carbohydrate recognition and transmembrane anion transport.
Carbohydrate Recognition
Binding carbohydrates from water is one 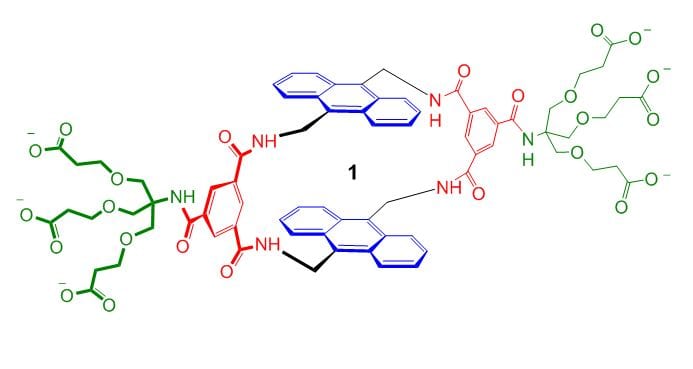 of the more difficult challenges in supramolecular chemistry. Carbohydrates are structurally complex and very hydrophilic, so prefer to stay in aqueous solution. Nonetheless, success can be achieved using cavities which are complementary to their substrates, deploying hydrogen bonding and apolar interactions in well-defined geometries. We have targeted the all-equatorial family of sugars (glucose and close relatives), partly because glucose-binding molecules can play a role in the management of diabetes. Recent developments are 1, a fluorescent glucose sensor; 2, a receptor for all-equatorial polysaccharides such as cellulose; and 3, a glucose receptor of extraordinary power and selectivity.
of the more difficult challenges in supramolecular chemistry. Carbohydrates are structurally complex and very hydrophilic, so prefer to stay in aqueous solution. Nonetheless, success can be achieved using cavities which are complementary to their substrates, deploying hydrogen bonding and apolar interactions in well-defined geometries. We have targeted the all-equatorial family of sugars (glucose and close relatives), partly because glucose-binding molecules can play a role in the management of diabetes. Recent developments are 1, a fluorescent glucose sensor; 2, a receptor for all-equatorial polysaccharides such as cellulose; and 3, a glucose receptor of extraordinary power and selectivity.
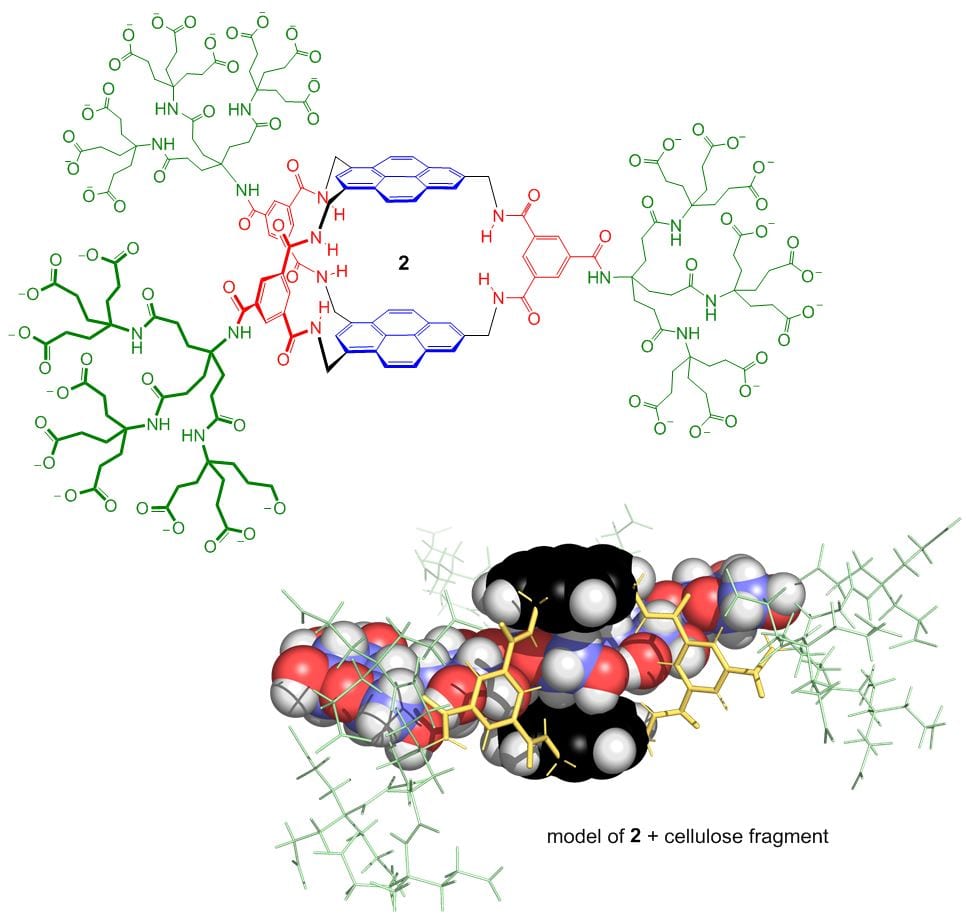
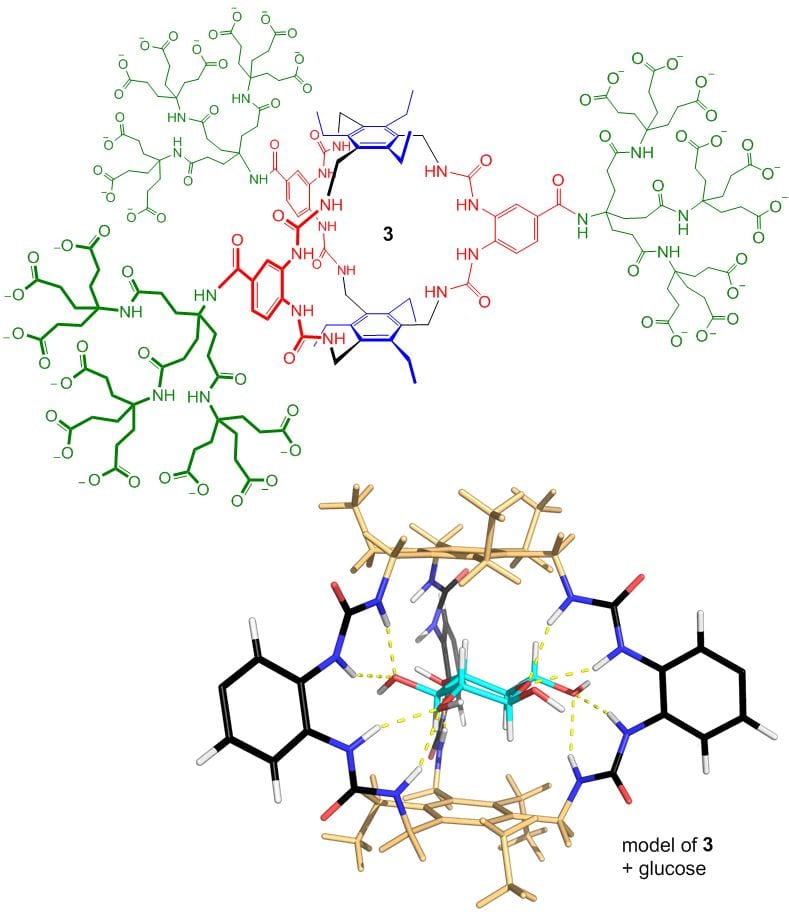
Receptor 3, otherwise known as “GluHUT” (Glucose-binding HexaUrea Temple) is especially significant. Its selectivity is almost perfect, allowing it to work in complex biological media. Its affinity for glucose matches all but the strongest carbohydrate-binding proteins. It works so well that medical applications are very realistic. For example, it could find use in insulin delivery systems and devices for monitoring blood glucose levels in diabetics. It has been commercialised through a start-up company, Ziylo, which was created to exploit the carbohydrate receptors designed in our group. In August 2018 Ziylo was sold to the Danish pharmaceutical company Novo Nordisk, in a deal that could be worth up to $800 million. Novo Nordisk have been using GluHUT to develop glucose-responsive insulin, in collaboration with our new company Carbometrics. Meanwhile research in our group is targeting other substrates, especially the many carbohydrates with axial substituents. From October 2023 this work, and other research inspired by GluHUT, will be funded by an ERC Advanced Grant providing over £2m of support.
Key references: Nature Chem. 2012, 4, 718-723; Nature Chem. 2016, 8, 69-74; Nature Chem. 2019, 11, 52-56; Chem. Soc. Rev. 2020, 49, 2531-2545.
Anion Transport across Biological Membranes
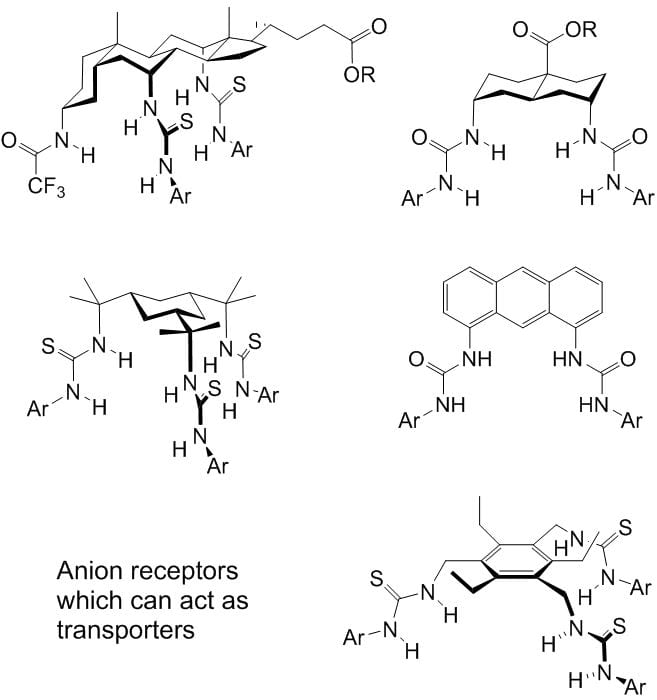
Transmembrane anion transport is an important biological process, normally occurring via protein channels. It is the absence or malfunction of one of the channels which causes the genetic disease cystic fibrosis (CF). We have designed a variety of powerful anion receptors which can locate in membranes, extract anions from water on one side and carry them across to the other. With collaborators in the School of Physiology and Pharmacology, and also in Sydney Australia, we are exploring their potential for “channel replacement therapy” to treat CF patients. In particular, we have found that some of our anion carriers (anionophores) are active in living cells, but show no sign of toxicity. We are encouraged to hope that delivery of the anionophores to the lungs of CF patients will be feasible, cause no harm, and replace the missing channel activity.
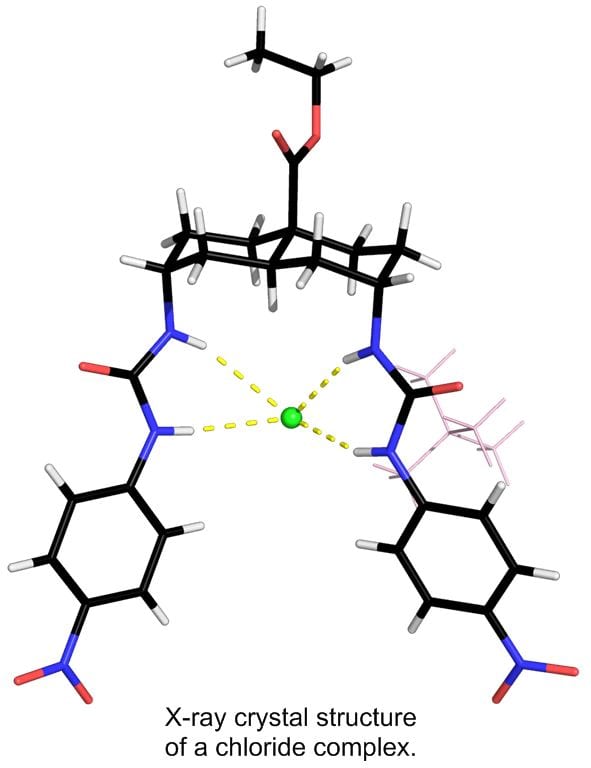
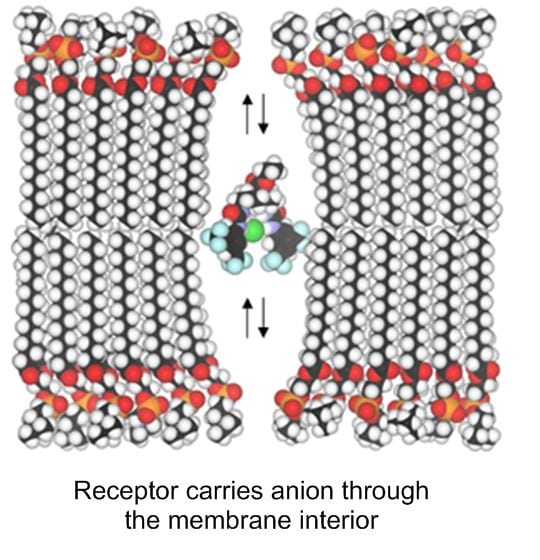
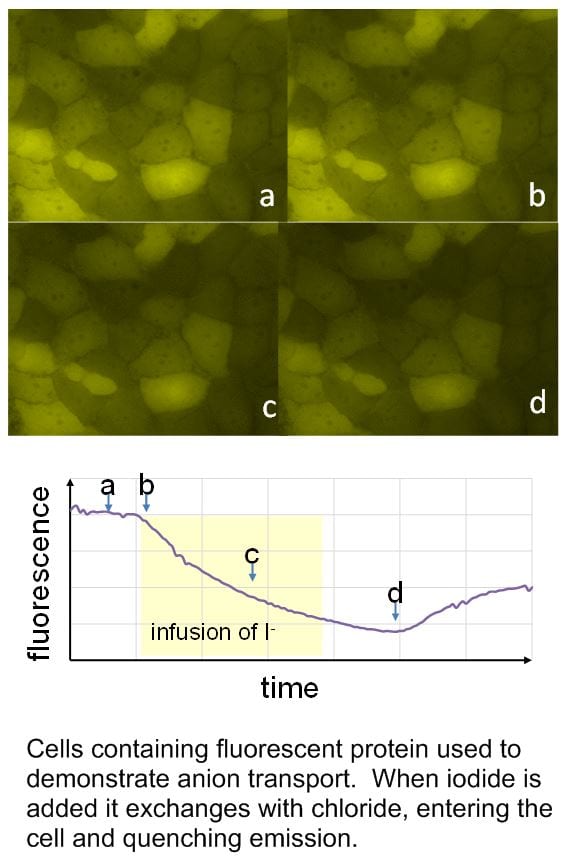
Key references: Acc. Chem. Res. 2013, 46, 2898-2909; Nature Chem. 2016, 8, 24-32; Chem 2016, 1, 127-146; Chem. Sci. 2019, 10, 9663-9672.