Synthetic Carbohydrate Receptors with Medical Applications
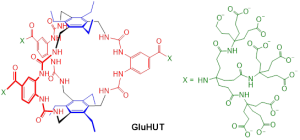
PhD studentships are available in the group of Prof. Tony Davis, to start October 2024. Projects will build on the remarkable success of GluHUT, the powerful and selective glucose-binding molecule which was reported recently by the group.1 As described under “research”, GluHUT has potential for various medical applications and was the subject of a commercial deal which could be worth as much as $800 million. Aims will be (a) to exploit GluHUT’s glucose binding properties in new ways which will further increase its value in medicine, and (b) to use the design as a starting point for receptors which bind other medically relevant carbohydrates. The projects will be supported by a collaboration with the group of Prof. A. J. Mulholland on state-of-art computational methodology for receptor design, including the use of virtual reality (VR) for building and manipulating candidate structures.
References
- A biomimetic receptor for glucose. R. A. Tromans, T. S. Carter, L. Chabanne, M. P. Crump, H. Li, J. V. Matlock, M. G. Orchard, A. P. Davis, Nature Chem. 2019, 11, DOI: 10.1038/s41557-018-0155-z . Biomimetic carbohydrate recognition. A. P. Davis, Chem. Soc. Rev. 2020, 49, 2531-2545, DOI: 10.1039/c1039cs00391f.
Application
Applicants must have obtained, or be about to obtain, a First or Upper Second Class UK first degree, or the equivalent qualifications gained outside the UK, in an appropriate area of science or technology. To make a formal application, follow the ’Start a new application’ link on this page: http://www.bristol.ac.uk/study/postgraduate/apply/. Please select Chemistry (PhD) on the Programme Choice page. You will be prompted to enter details of the studentship in the Funding and Research Details sections of the form. We encourage you to make an informal enquiry to Prof Davis (Anthony.Davis@bristol.ac.uk) if you have any queries or would like to discuss projects.
Funding Notes
A full studentship will cover tuition fees, a training support fee and a stipend at standard EPSRC rates for 4 years.